New drug delivery method can reverse senescence of stem cells
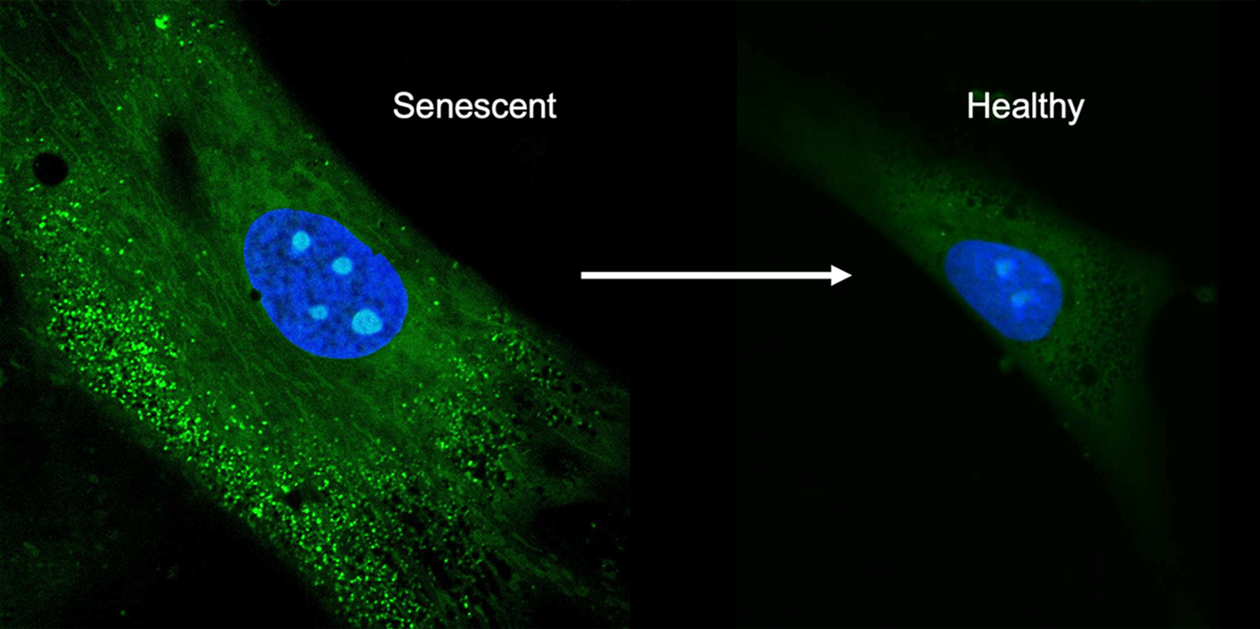
Confocal images of mesenchymal stem cells. The left shows the senescent cells producing unwanted biomolecules, the right shows the cells after treatment with the antioxidant crystals. / Kong lab
As we age, our bodies change and degenerate over time in a process called senescence. Stem cells, which have the unique ability to change into other cell types, also experience senescence, which presents an issue when trying to maintain cell cultures for therapeutic use. The biomolecules produced by these cell cultures are important for various medicines and treatments, but once the cells enter a senescent state they stop producing them, and worse, they instead produce biomolecules antagonistic to these therapeutics.
While there are methods to remove older cells in a culture, the capture rate is low. Instead of removing older cells, preventing the cells from entering senescence in the first place is a better strategy, according to Ryan Miller, a postdoctoral fellow in the lab of Hyunjoon Kong (M-CELS leader/EIRH/RBTE), a professor of chemical and biomolecular engineering.
“We work with mesenchymal stem cells, that are derived from fat tissue, and produce biomolecules that are essential for therapeutics, so we want to keep the cell cultures healthy. In a clinical setting, the ideal way to prevent senescence would be to condition the environment that these stem cells are in, to control the oxidative state,” said Miller. “With antioxidants, you can pull them the cells out of this senescent state and make them behave like a healthy stem cell.”
While treating the cells with antioxidants can delay senescence, current methods of antioxidant delivery have many shortcomings, including large variation in amount of drug release over time and between cells. However, a recently published study by the labs of Kong and Hee-Sun Han (GNDP/IGOH), an assistant professor of chemistry, with Miller as first author, describes a new method of delivering antioxidants to stem cells that is reliable, long-lasting, and minimizes variation.
The new method utilizes antioxidants in the form of polymer-stabilized crystals. Traditional methods grow crystals within reactors, but using microfluidics, a technology that allows researchers to work with incredibly small amounts of fluid, the researchers can create crystals that are all the same size and dosage, minimizing variation in drug release between cells. “With microfluidics, each drop functions as a small reactor, such that we can get small, similar-sized, individual crystals, which minimizes variation in drug release rate.” said Miller. Furthermore, the crystals dissolve at a slower rate than traditional methods, making the release of the drug uniform over time, and increasing the duration of the drug’s effectiveness.
“We learned that the narrow variation in the drug’s release profile is really important,” explained Han. “When you add drugs that dissolve in water, there is this bursting period where a lot of is dissolves in the liquid at once, and not much later. But the crystal allows this uniform, extended release, which helps maintain the tight range of optimal concentrations that are needed.”
“When typical antioxidants are put into water or biological fluid, they lose their vital activity within six hours,” described Kong. “But the new antioxidant crystal remains bioactive for at least two days, so we can actually extend the duration of the drug, and also reduce the frequency with which we have to add antioxidants to the cell culture media. This minimizes the variation in the type of the biomolecules the stem cells are generating and improves the reproducibility of the product, which is one of the biggest challenges in biomanufacturing at the moment.”
Increased duration of the drug’s efficacy means that stem cell cultures can be kept out of the senescence state for longer, which leads to a larger harvest of the needed biomolecules for therapeutics. Miller also says this method could be used for patient-derived stem cell treatments, where the biomolecules from a patient’s own body are used to help with various tissue ailments, such as injuries or disease.
“When we use biomolecules from donors instead of the patient, that can have a host effect,” explained Miller. “Ideally, we would harvest stem cells from the patient that we're treating, grow them in a bioreactor, and harvest those biomolecules for that therapeutic. This works well for someone who is 20, but if we envision an elderly patient, they’re going to have a high population of these senescence cells, that are not going to be secreting the therapeutically relevant biomolecules. If we can pull those cells out of that state, and make them behave like a healthy cell, we can get a much larger load of therapeutically relevant biomolecules for the patient.”
The team says that while they want to continue to improve the biomanufacturing process, there are already many potential uses for this methodology besides just controlled delivery of antioxidants to stem cell cultures. Most cells experience senescence, so this technique could be applied to other cell cultures important in medicine and therapeutics. Furthermore, the crystals could be used to deliver sustained and controlled levels of antioxidants, or potentially other drugs, directly into the target tissue of a patient.
“I think the beauty here is that this is a technology development paper, so this can be applied to various hydrophilic drugs, disease models, and methods applications,” said Han. “We’re showing that we can maintain a sustained release of this drug at a relatively constant rate for an extended period of time. There are a lot of exciting studies and directions that we can go with this technology.”
The study is published in Advanced Functional Materials and can be found at https://doi.org/10.1002/adfm.202302232
